Driving Question:
How could Nanoscience affect the quality of our drinking water?
|
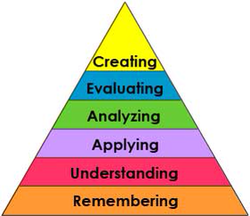
Our learning objectives are designed to span Bloom's hierarchy of higher-order thinking skills. The numbers listed next to the learning objectives that follow correspond to the higher-order thinking skill from Bloom's Taxonomy being incorporated from the list below:
|
Learning Objectives:
Overarching Learning Objective:
Learners will be able to create a solution to control the quality of drinking water (6)
Nanoscience Learning Objectives:
Learners will be able to...
Chemistry Learning Objectives:
Learners will be able to...
Back to top
Learners will be able to create a solution to control the quality of drinking water (6)
Nanoscience Learning Objectives:
Learners will be able to...
- 1.1 Relate the size of nano-sized objects to objects encountered in daily life (4)
- 1.2 Compare surface-area-to-volume ratios of different sized objects and explain how surface-area-to-volume ratios play a role in properties of objects at the nanoscale (4)
- 1.3 Explain why properties of matter can change at the nano scale (3)
- 1.4 Apply dimensional analysis in conversion problems (3)
Chemistry Learning Objectives:
Learners will be able to...
- Analyze water samples to determine ions and chemicals present (4)
- Calculate the concentration of different chemicals in water using the molarity concept (2)
- Explain the properties of mixtures and solutions and classify different samples using chemical and physical properties (2)
- Prepare solutions that contain desired amounts of chemical components (3)
Back to top
Indiana Academic Standards
Chemistry I Standards:
C.1.1 - Based on physical properties, differentiate between pure substances and mixtures.
C.1.2 - Observe and describe chemical and physical properties of different types of matter and designate them as either extensive or intensive.
C.3.1- Describe, compare and contrast the characteristics of the interactions between atoms in ionic and covalent compounds.
C.3.2- Compare and contrast how ionic and covalent compounds form.
C.4.7- Perform calculations to determine the percent composition of a compound or mixture when given a formula.
C.6.1- Explain that atoms and molecules are in constant motion and that this motion increases as thermal energy increases.
C.7.1 - Describe the composition and properties of types of solutions.
C.7.2 - Explain how temperature, pressure and polarity of the solvent affect the solubility of a solute.
C.7.3 - Describe the concentration of solutes in a solution in terms of molarity. Perform calculations using molarity, mass and volume.
C.7.4 - Prepare a specific volume of a solution of a given molarity when provided with a known solute.
C.7.5 - Explain how the rate of a reaction is qualitatively affected by changes in concentration, temperature, surface area and the use of a catalyst.
Back to top
C.1.1 - Based on physical properties, differentiate between pure substances and mixtures.
C.1.2 - Observe and describe chemical and physical properties of different types of matter and designate them as either extensive or intensive.
C.3.1- Describe, compare and contrast the characteristics of the interactions between atoms in ionic and covalent compounds.
C.3.2- Compare and contrast how ionic and covalent compounds form.
C.4.7- Perform calculations to determine the percent composition of a compound or mixture when given a formula.
C.6.1- Explain that atoms and molecules are in constant motion and that this motion increases as thermal energy increases.
C.7.1 - Describe the composition and properties of types of solutions.
C.7.2 - Explain how temperature, pressure and polarity of the solvent affect the solubility of a solute.
C.7.3 - Describe the concentration of solutes in a solution in terms of molarity. Perform calculations using molarity, mass and volume.
C.7.4 - Prepare a specific volume of a solution of a given molarity when provided with a known solute.
C.7.5 - Explain how the rate of a reaction is qualitatively affected by changes in concentration, temperature, surface area and the use of a catalyst.
Back to top
Next Generation Science Standards
High School Physical Science Standards:
HS-PS2-6 - Communicate scientific and technical information about why the molecular-level structure is important in the functioning of designed materials.
HS-PS3-5 - Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the forces between objects and the changes in energy of the objects due to the interaction.
For other disciplinary NGSS standards, please click here.
Back to top
HS-PS2-6 - Communicate scientific and technical information about why the molecular-level structure is important in the functioning of designed materials.
HS-PS3-5 - Develop and use a model of two objects interacting through electric or magnetic fields to illustrate the forces between objects and the changes in energy of the objects due to the interaction.
For other disciplinary NGSS standards, please click here.
Back to top
Classroom Culture and Leadership Skills
21st Century Skills:
- Think critically to collaborate with group members.
- Creatively innovate and construct a possible solution to a global problem.
- Use information, media, and technology to acquire and utilize information.
- Develop and practice leadership skills such as responsibility, flexibility, adaptability, initiative, self direction, accountability and productivit
Detailed Plan for Communicating Standards to Students & Sample Materials
The teacher will:
Back to top
- Post the standards at the beginning of the unit on the Learning Management System (LMS), such as Canvas or Blackboard, as well as on the classroom on the wall. View the learning objectives that will be posted in the classroom here.
- Post clear and simple objectives prior to the start of the project on the LMS, teacher webpage, and the wall or board. See link above.
- Go over the final project rubric after the entry event captures the student's interest to keep them focused on the standards and objectives expected of them to cover for the duration of the unit.
- Show students sample materials from student projects from previous years and/or examples from the internet or learning community.
Back to top
Detailed Plan for Ensuring Students Can Explain Their Learning
Students will:
Resource: Bloom's Taxonomy. (n.d.). Retrieved June 10, 2015, from http://ww2.odu.edu/educ/roverbau/Bloom/blooms_taxonomy.htm
Resource: Framework for 21st Century Learning - P21. (n.d.). Retrieved June 11, 2015, from http://www.p21.org/our-work/p21-framework
Back to top
- Complete exit tickets throughout the unit to ensure students can relate what they learned to expected learning objectives
- Write journal reflections in which they indicate what they learned or still have questions about
- Analyze the content in activities and laboratories to ensure they have learned the proper content
- Take both a pre- and post- assessments to ensure they have learned the content from the unit
Resource: Bloom's Taxonomy. (n.d.). Retrieved June 10, 2015, from http://ww2.odu.edu/educ/roverbau/Bloom/blooms_taxonomy.htm
Resource: Framework for 21st Century Learning - P21. (n.d.). Retrieved June 11, 2015, from http://www.p21.org/our-work/p21-framework
Back to top